Birth Control Pills Could Soon Be Available Without A Prescription
The FDA has a few questions to answer before arriving at its decision.
News that is entertaining to read
Subscribe for free to get more stories like this directly to your inboxThe issue of women’s reproductive choices has been at the forefront of American political discussion for a while now — and this week, the Food and Drug Administration provided what some will see as positive news on that front.
According to reports, the agency is considering whether birth control pills should be available without a prescription, removing one barrier between women and contraception.
Digging into the debate
An advisory panel within the FDA began a two-day meeting yesterday with the goal of determining whether the evidence supports removing the need for a doctor’s approval.
While it’s not difficult for many women to receive the pill, women’s health advocate Kelly Blanchard said that others lack easy access to a healthcare provider and face other hurdles that prevent them from obtaining a prescription.
A number of prominent medical organizations support the push to offer a progestin-only pill to be offered over the counter
The proposal is facing backlash over fears that, among other things, sex traffickers could use it to more easily exploit victims.
Asking the right questions
It is the FDA panel’s job to cover all of the possible downsides of removing the need for a prescription, such as determining how women would know when and how to take the pill as well as whether any health issues would interact with the medication if a doctor is no longer involved.
Dr. Timothy Millea of the Catholic Medical Association has voiced his concern that removing the need for a doctor’s visit might “eliminate the screenings for ovarian cancer, for cervical cancer, for sexually transmitted infections.”
Dozens of other countries already offer the pill without a prescription, but U.S. regulators will need to weigh all the pros and cons before issuing their decision, which is expected to be announced by the end of summer.
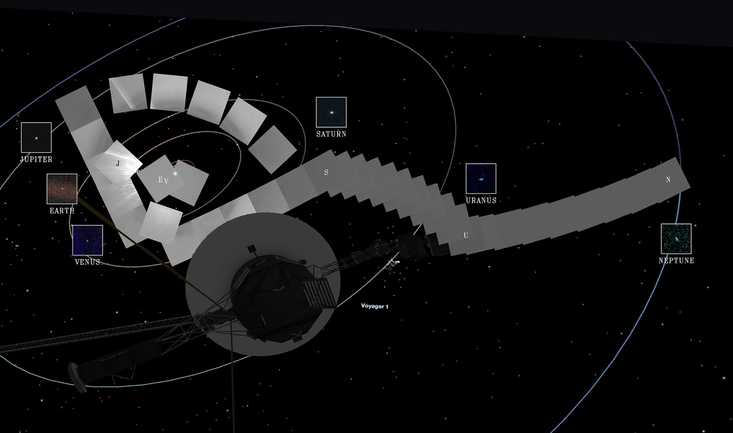 Why Is The Aging Voyager 1 Probe Sending Back Incoherent Communications?
It's been speaking gibberish for a few months and officials are concerned.
Why Is The Aging Voyager 1 Probe Sending Back Incoherent Communications?
It's been speaking gibberish for a few months and officials are concerned. One Woman’s Massive Donation Is Wiping Out Tuition At This Medical School
Her inheritance came with the instruction to do "whatever you think is right."
One Woman’s Massive Donation Is Wiping Out Tuition At This Medical School
Her inheritance came with the instruction to do "whatever you think is right." Woman’s Pets Will Inherit Her Multimillion-Dollar Fortune, Not Her Kids
It's not the first time four-legged heirs were named in a will.
Woman’s Pets Will Inherit Her Multimillion-Dollar Fortune, Not Her Kids
It's not the first time four-legged heirs were named in a will.