How A Rebrand Helped This Chinese Vape Company Evade US Regulators
The FDA might be cracking down, but there are some serious shortcomings. Shutterstock
Shutterstock
News that is entertaining to read
Subscribe for free to get more stories like this directly to your inbox“What’s in a name?” It’s a question Juliet asked in the timeless Shakespeare tragedy and countless others have pondered in the centuries since.
Well, one Chinese e-cigarette company has clearly figured out that a name means a lot — especially when you’re trying to avoid having your product confiscated by U.S. authorities.
Illegal vapes
American regulators have cracked down on various types of vaping products in recent years, including the fruit-flavored devices marketed by the Chinese company known in the States as Elf Bar.
But when the Food and Drug Administration tasked customs officials with confiscating these products upon arrival, the company wasn’t willing to just chalk it up as a loss. Instead, it took the simple, but ultimately pretty effective, step of just changing its name.
Elf Bar became known as EBCreate and the manufacturers listed on its packaging also received a new name to avoid being linked to the names on the FDA’s off-limits list.
Surging sales
While Elf Bar’s tactics might seem brazen, this is hardly the only e-cigarette company that has tried to get over on the FDA (and succeeded).
That’s why the Public Health Law Center is pressuring the FDA to do a better job in its role as a regulating force in the industry. Specifically, the group advises the implementation of unique ID codes on any product that falls under the agency’s purview.
In just the few years since 2020, estimates indicate that the number of individual e-cig brands available on U.S. shelves has tripled, and most of them originate in China. This makes the FDA’s job much harder, but the agency’s tobacco director said that there is “a variety of tools at our disposal to take action” against companies like Elf Bar that try to pull a fast one.
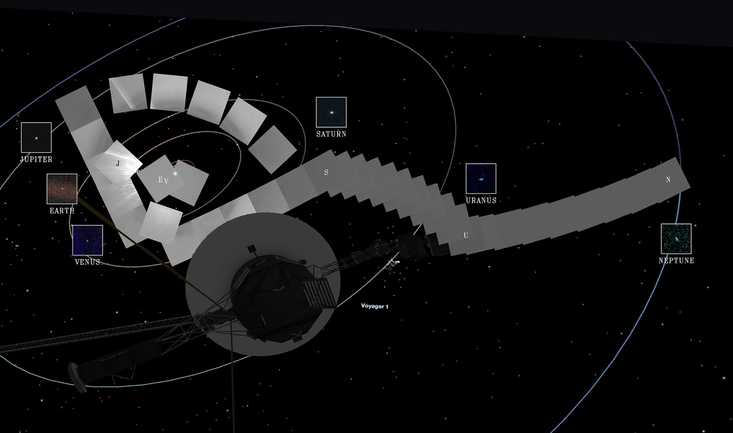 Why Is The Aging Voyager 1 Probe Sending Back Incoherent Communications?
It's been speaking gibberish for a few months and officials are concerned.
Why Is The Aging Voyager 1 Probe Sending Back Incoherent Communications?
It's been speaking gibberish for a few months and officials are concerned. One Woman’s Massive Donation Is Wiping Out Tuition At This Medical School
Her inheritance came with the instruction to do "whatever you think is right."
One Woman’s Massive Donation Is Wiping Out Tuition At This Medical School
Her inheritance came with the instruction to do "whatever you think is right." Woman’s Pets Will Inherit Her Multimillion-Dollar Fortune, Not Her Kids
It's not the first time four-legged heirs were named in a will.
Woman’s Pets Will Inherit Her Multimillion-Dollar Fortune, Not Her Kids
It's not the first time four-legged heirs were named in a will.